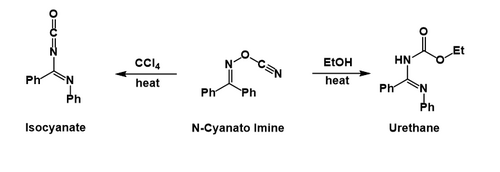
PDF) Manganese triacetate mediated regeneration of carbonyl compounds from oximes | Cihangir Tanyeli - Academia.edu

Mechanistic Insight into Self-Propagation of Organo-Mediated Beckmann Rearrangement: A Combined Experimental and Computational Study | The Journal of Organic Chemistry

PDF) Nitric Acid and Phosphorus Pentoxide Supported on Silica Gel as a Mild and Efficient System for the Oxidation of Benzylic Alcohols under Solvent-Free Conditions | Arnold Ruoho - Academia.edu

Copper-Catalyzed Unstrained C–C Single Bond Cleavage of Acyclic Oxime Acetates Using Air: An Internal Oxidant-Triggered Strategy toward Nitriles and Ketones | The Journal of Organic Chemistry

Efficient synthesis of carbonyl compounds: oxidation of azides and alcohols catalyzed by vanadium pentoxide in water using tert-butylhydroperoxide - ScienceDirect
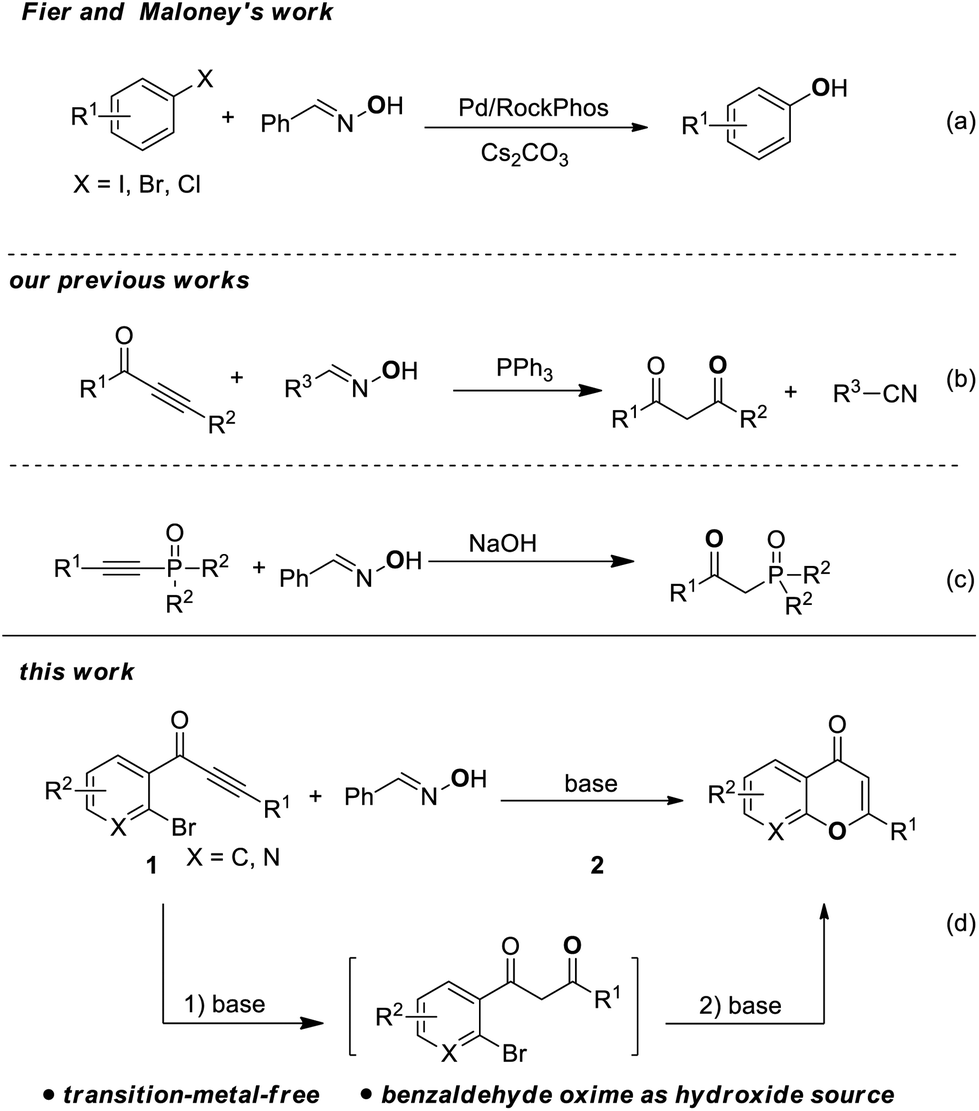
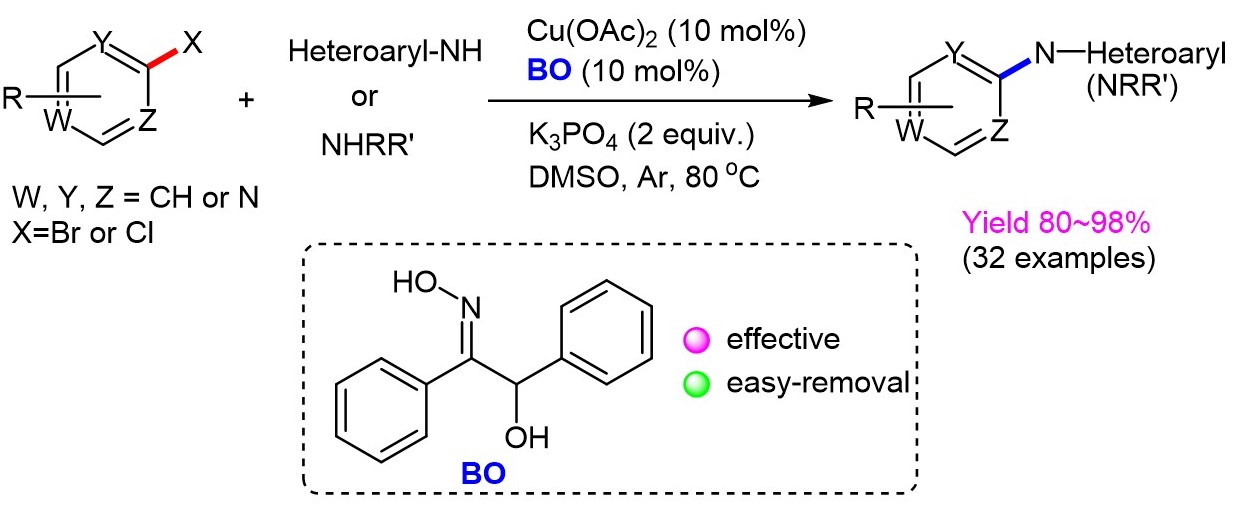
An efficient tandem synthesis of chromones from o -bromoaryl ynones and benzaldehyde oxime - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB01387C

a) Synthesis of the amidoxime‐functionalized PAO. b) The XPS spectra of... | Download Scientific Diagram

Enantioselective reduction of ketoxime ethers with borane–oxazaborolidines and synthesis of the key intermediate leading to (S)-rivastigmine - ScienceDirect
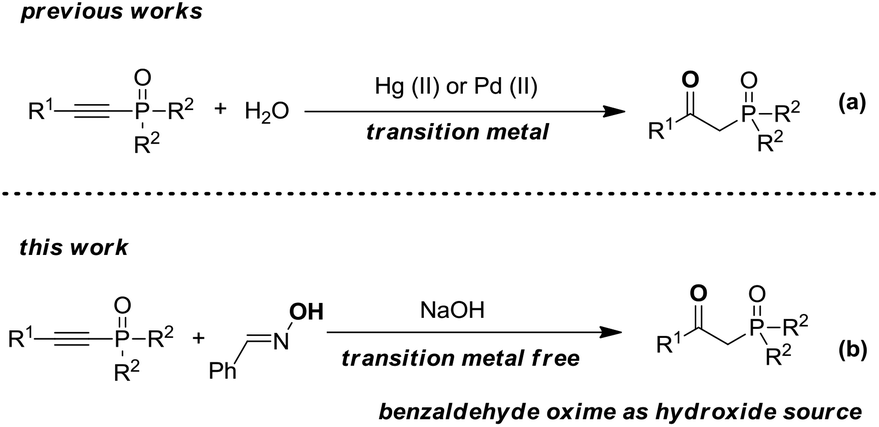
An efficient preparation of β-ketophosphine oxides from alkynylphosphine oxides with benzaldehyde oxime as a hydroxide source - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB00251K

The Mechanochemical Beckmann Rearrangement over Solid Acids: From the Ball Mill to the Extruder - Baier - Chemistry–Methods - Wiley Online Library


![Synthesis of 2-amino-2'-fluoro-5-[1-(hydroxyimino)ethyl]benzophenone oxime Synthesis of 2-amino-2'-fluoro-5-[1-(hydroxyimino)ethyl]benzophenone oxime](https://prepchem.com/image/substance/627hprn3wvwgs4owswgoss40g8ssc8cs4owkggooo048g8ws4o)