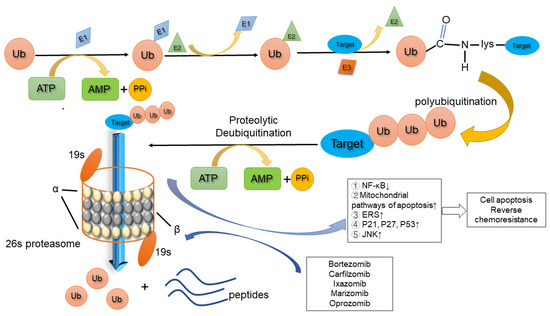
Sp. zn. sukls75235/2019 SOUHRN ÚDAJŮ O PŘÍPRAVKU 1. NÁZEV PŘÍPRAVKU Bortezomib Teva 3,5 mg prášek pro injekční roztok

Teva Announces Launch of a First Generic Version of Revlimid® (lenalidomide capsules), in the U.S. | Business Wire
Package leaflet: Information for the user Bortezomib Teva 2.5 mg powder for solution for injection Bortezomib Read all of this l
View of Stability of Generic Formulations of Bortezomib 1.0 and 2.5 mg/mL in Vials and Syringes Stored at 4°C and Room Temperature (23°C or 25°C) | Canadian Journal of Hospital Pharmacy
1 de 37 FICHA TÉCNICA 1. NOMBRE DEL MEDICAMENTO Bortezomib Teva 2,5 mg polvo para solución inyectable 2. COMPOSICIÓN CUALITA
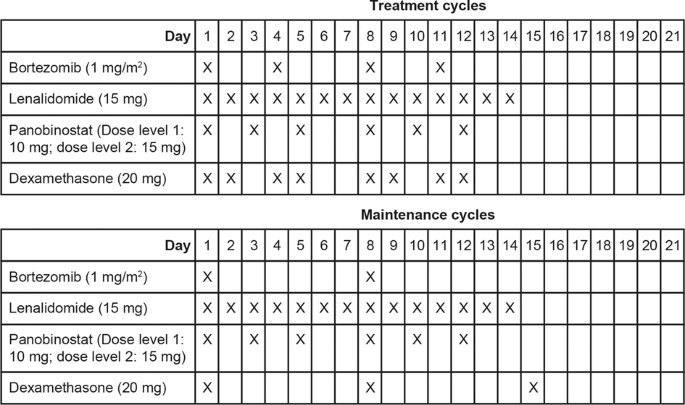
Phase 1 open-label study of panobinostat, lenalidomide, bortezomib + dexamethasone in relapsed and relapsed/refractory multiple myeloma | Blood Cancer Journal
Sp. zn. sukls75235/2019 Příbalová informace: informace pro uživatele Bortezomib Teva 2,5 mg prášek pro injekční roztok b

Efficacy and safety of oral panobinostat plus subcutaneous bortezomib and oral dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma (PANORAMA 3): an open-label, randomised, phase 2 study - The