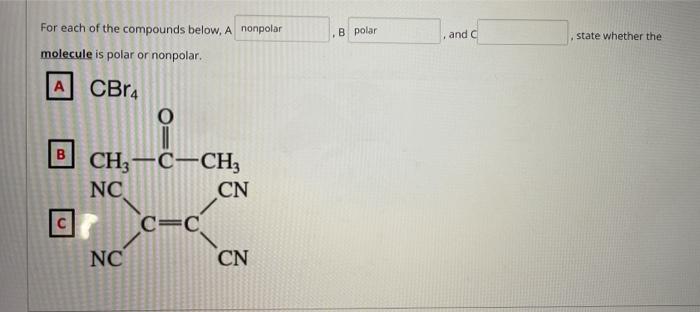
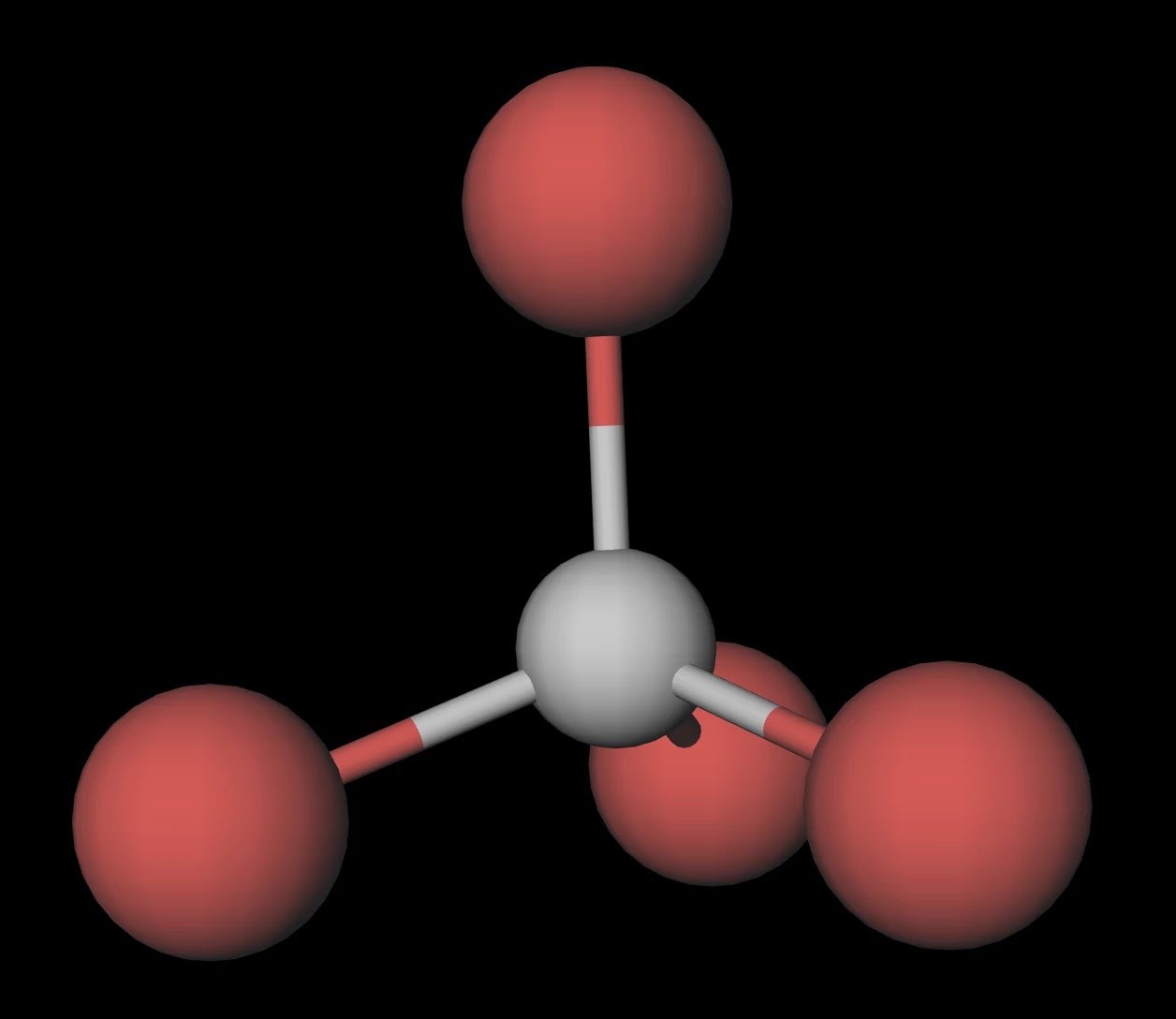
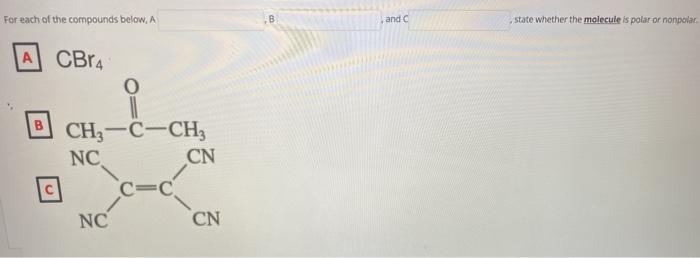
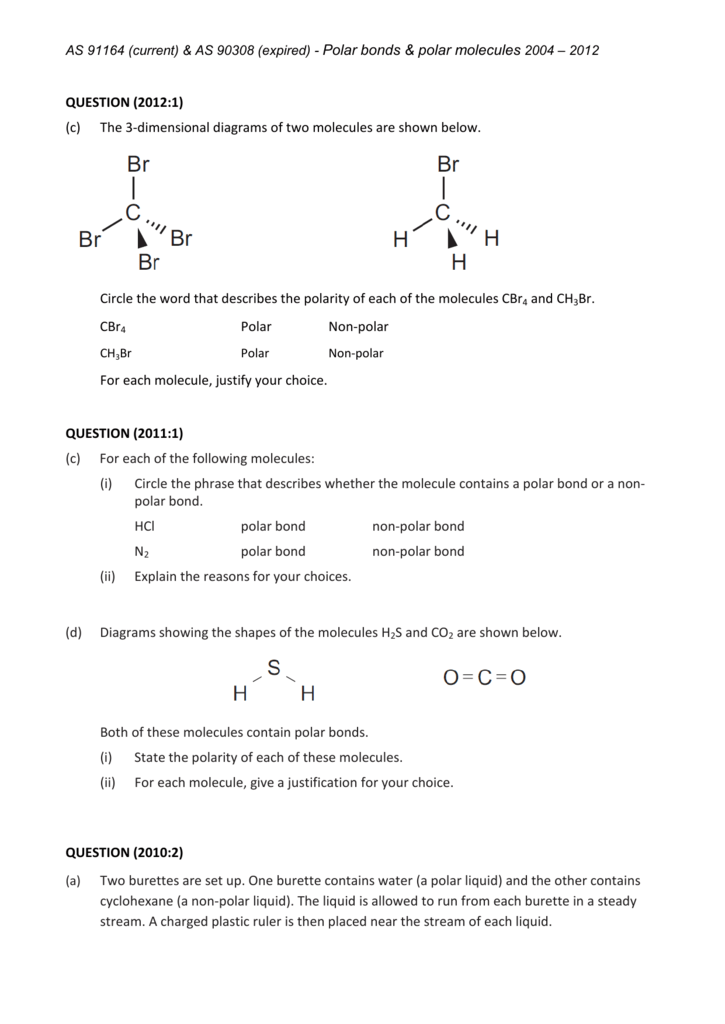
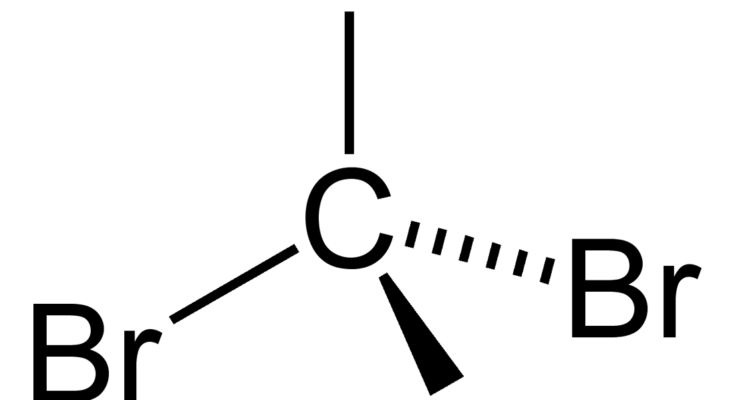
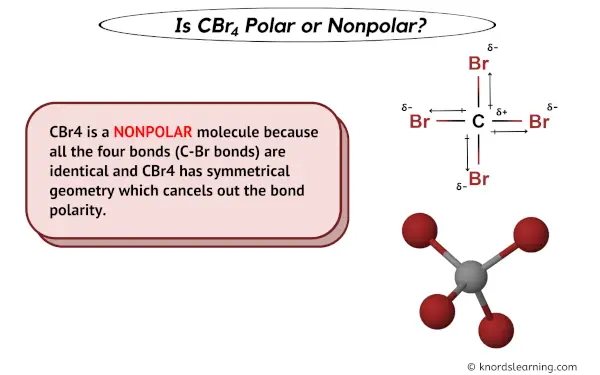
The carbon tetrabromide molecule, CBr4, is A a polar molecule with polar bonds B a nonpolar molec - YouTube

SOLVED: In which of the following solvents will CBr4 have the greatest solubility? CH3OH CH3Br H2C=O C8H18

Provide the following information for the compound CBr4. a. number of shared electron pairs b. number of unshared electron pairs c. shape of molecule d. ionic, polar covalent, or nonpolar covalent