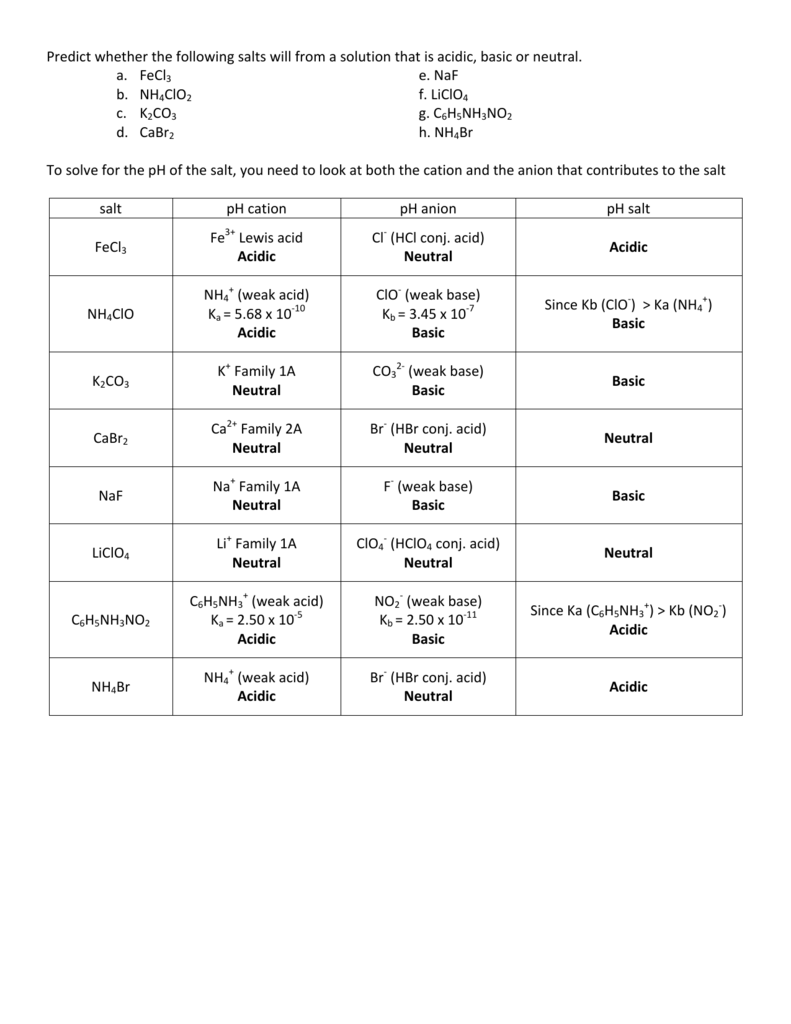
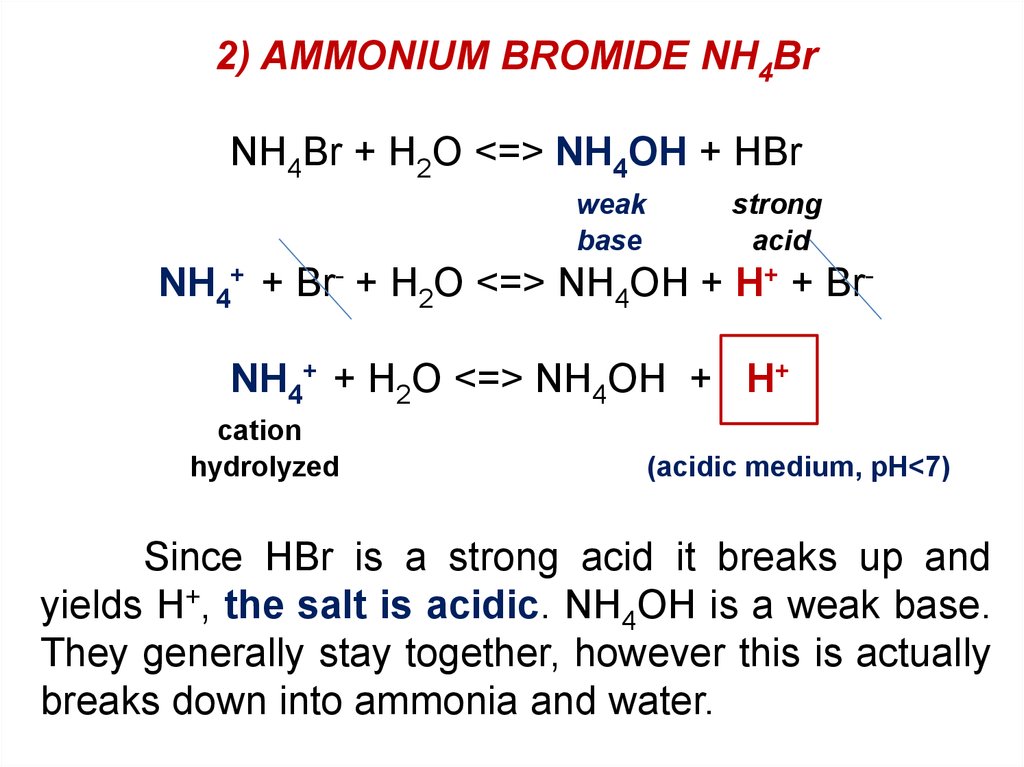
Which salt produces a basic aqueous solution at 25 degrees Celsius? a. KBr b. NH4Br c. NaF d. LiI e. BaCl2 | Homework.Study.com

SOLVED:Classify each of the following salt solutions as acidic, basic, or neutral: (a) KBr (b) NaNO (c) NH4Br (d) ZnCl (e) NH4F

SOLVED: Predict whether aqueous solutions of the following compounds are acidic, basic, or neutral compound solution is .? acidic A(C1o4) , basic neutral acidic NH4Br basic neutral acidic K Br basic neutral

NH4Br --> acid LiNO3 --> neutral K2SO3 --> basic KCl --> neutral Na2S --> basic - Home Work Help - Learn CBSE Forum