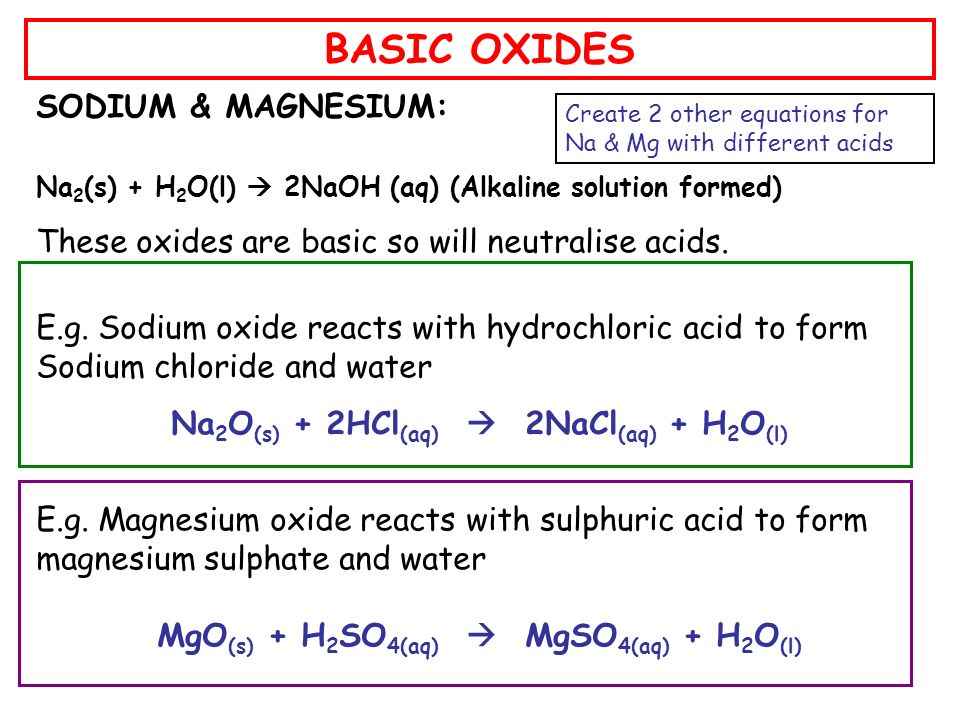
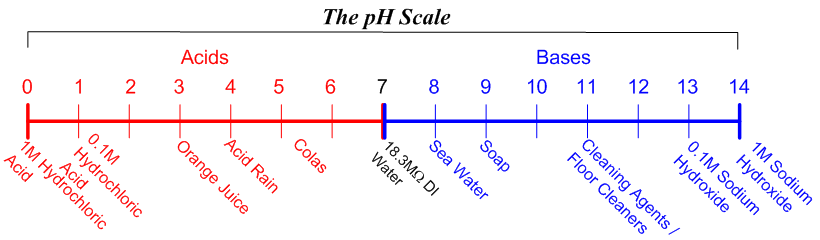
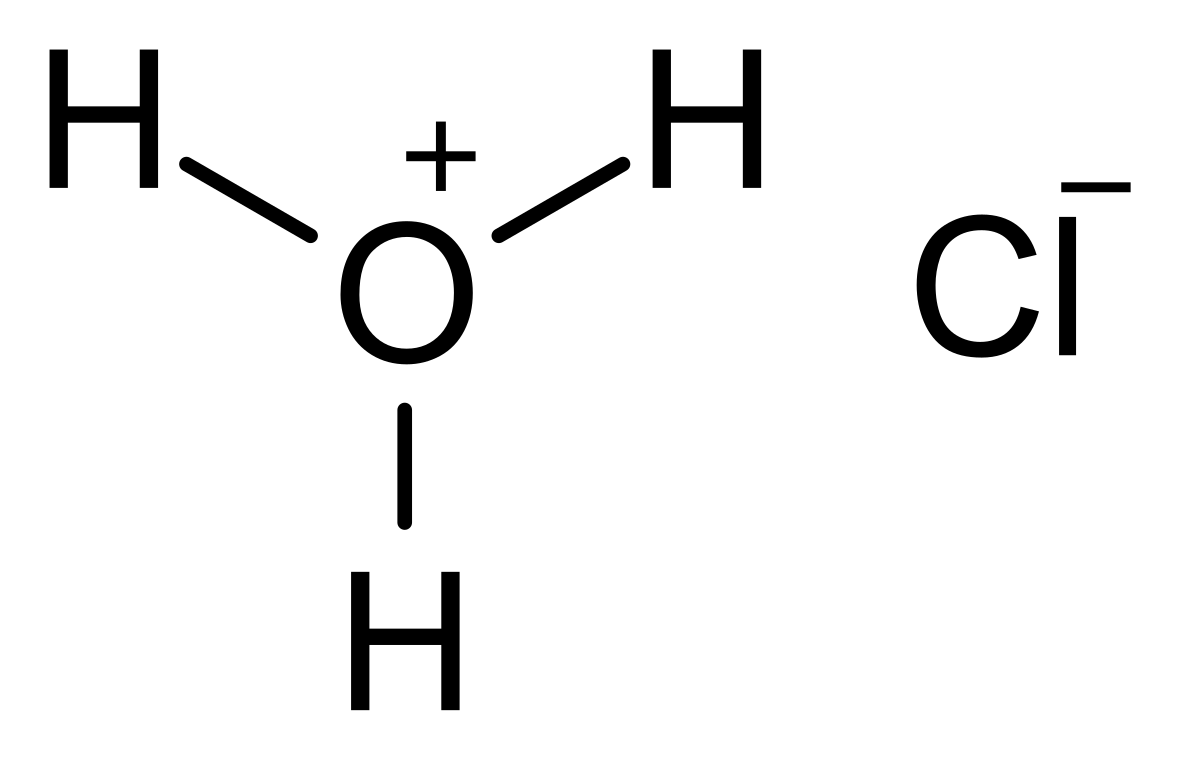
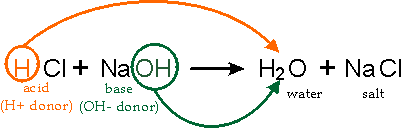
Write a balanced chemical equation when sodium reacts with a hydrochloric acid to produce sodium chloride solution and water.

H 2 SO 4 + Zn 1) Sulphuric acid + zinc 3) Nitric acid + sodium thiosulphate 2) Hydrochloric acid + magnesium 4) Hydrogen peroxide with catalyst Popular. - ppt download
The Solubility of Arsenious Oxide in Dilute Solutions of Hydrochloric Acid and Sodium Hydroxide. The Character of the Ions of Trivalent Arsenic. Evidence for Polymerization of Arsenious Acid | Journal of the
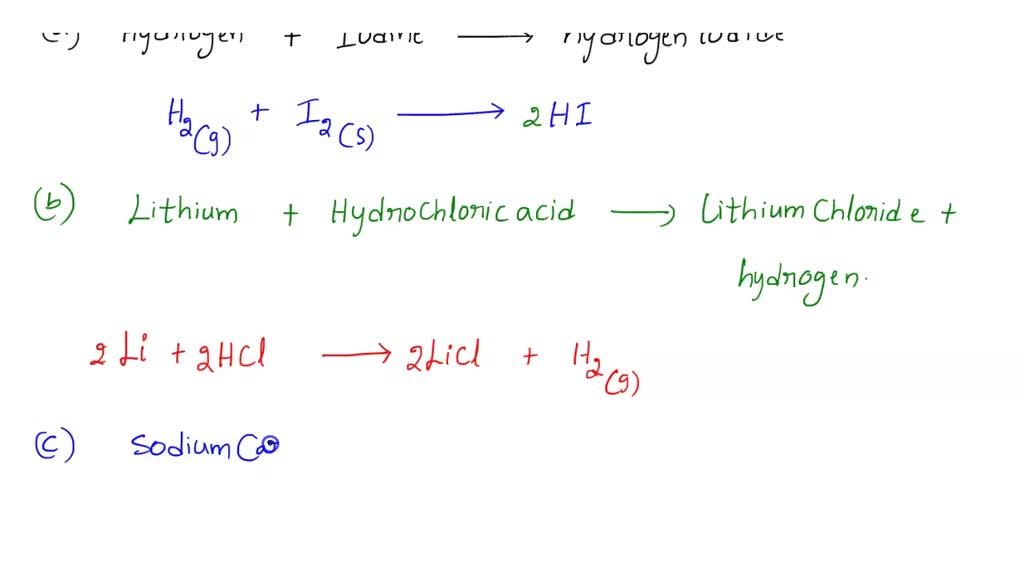
SOLVED: Write and balance each of the following equations, and then identify each by type. a. hydrogen + iodine ⟶ hydrogen iodide b. lithium + hydrochloric acid ⟶ lithium chloride + hydrogen

Sodium Oxide (Na2O) - Structure, Physical Properties, Chemical Properties and Uses with FAQs of Sodium Oxide

How would you write the name of the following compounds ◦ Zn(OH) 2 ◦ NaOH ◦ HCl ◦ Mg(NO 3 ) 2 What does an acid do to red litmus? What does an acid to. - ppt download
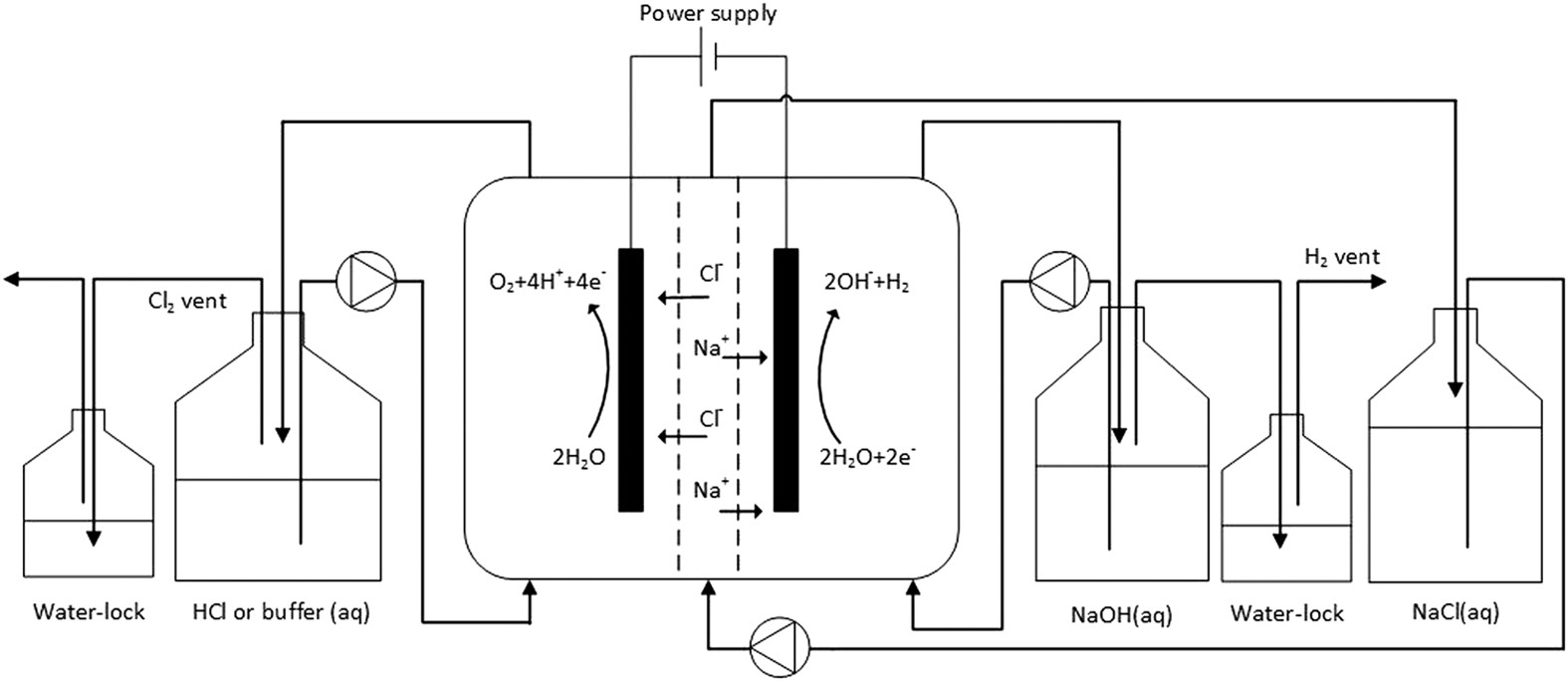
Direct anodic hydrochloric acid and cathodic caustic production during water electrolysis | Scientific Reports

Give the balanced equation for action of hydrochloric acid on lead dioxide - Chemistry - Analytical Chemistry Uses of Ammonium Hydroxide and Sodium Hydroxide - 12264393 | Meritnation.com