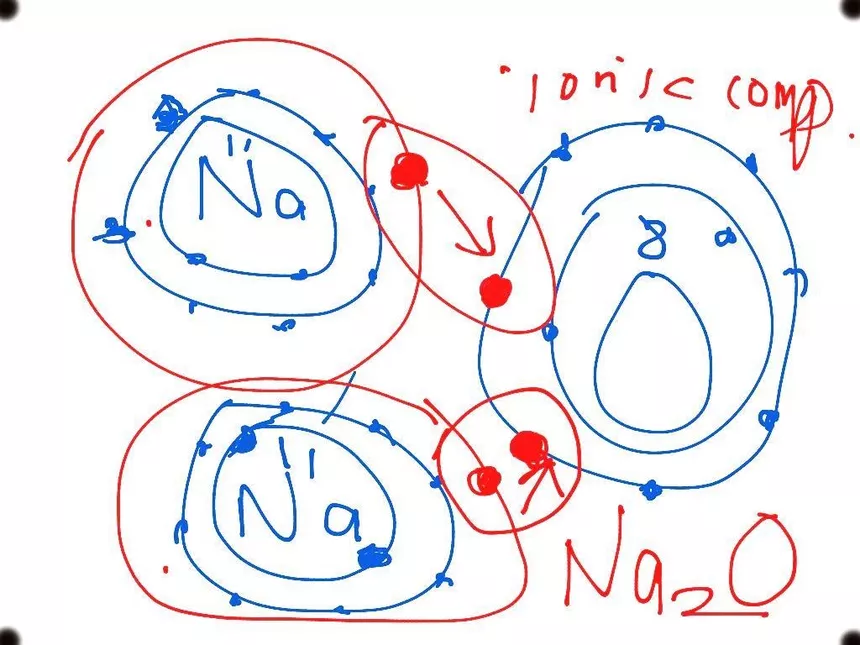
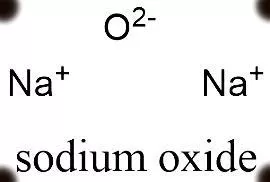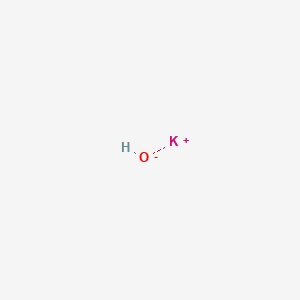This question is about sodium and oxygen to form sodium oxide. Sodium Oxide Contains sodium Ions (NA *) and Oxide Ions (0²-). Describe, in terms of electrons, what happens help sodium atoms

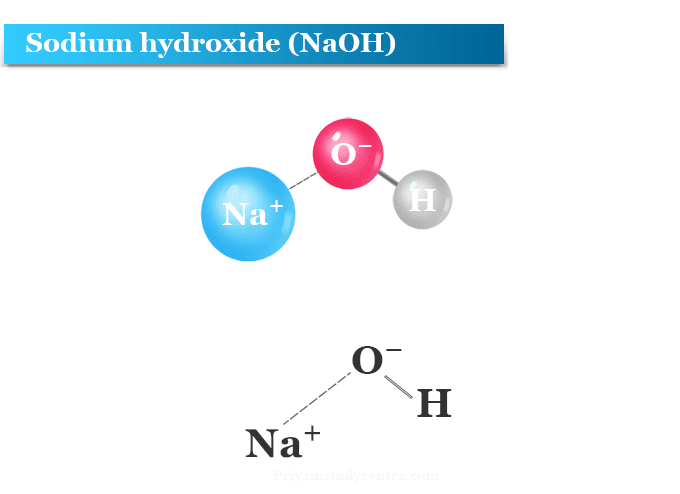
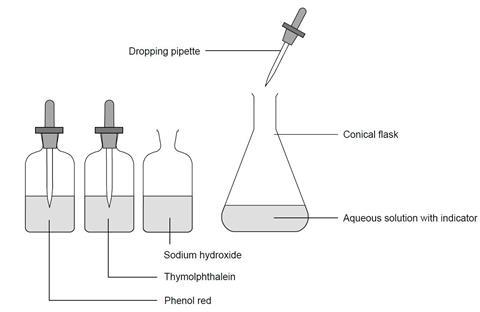
For students A,B,C and D were studying the effect of the solutions of hydrochloric acid, sodium hydroxide, sodium chloride and pure water respectively on the blue litmus solutioin. Which of the students

Sodium Oxide (Na2O) - Structure, Physical Properties, Chemical Properties and Uses with FAQs of Sodium Oxide